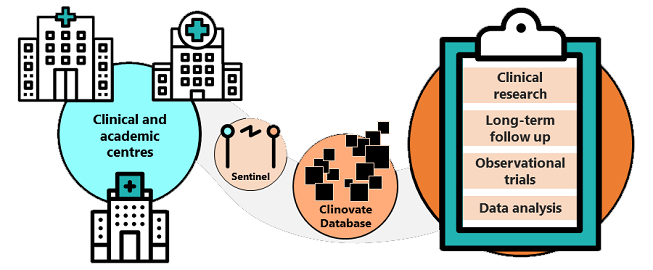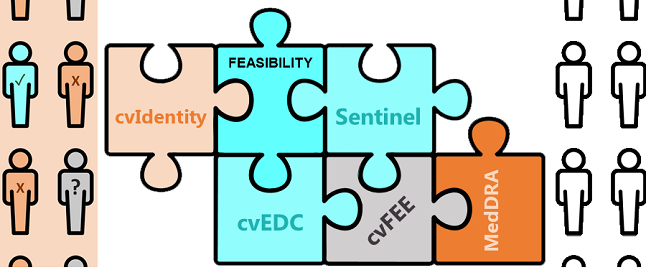
cvFeasibility - Increase success!
Clinovate NET's cvEDC (Electronic Data Capture) is a clinical data capture and management solution for a variety of research needs. We focus on designing our EDC to meet a variety of needs of collection sites and research groups.
Our solution provides a user-friendly, browser-based, responsive interface through which your data is easily and securely documented, protected, and backed up. In managing our data collection solutions, we adhere to FDA CFR Part 11 and GCP guidelines.
We strive to make the processing, review and control of this information as user-friendly as possible by appropriately flagging, recording and labeling each incident for easy review and resolution.